Update Vaccine-Associated CVT | CNSF Members
from the Canadian Stroke Consortium
VACCINE-ASSOCIATED CEREBRAL VENOUS THROMBOSIS
8 May 2021
|
Summary: - There continue to be rare instances of Vaccine-induced Thrombotic Thrombocytopenia (VITT), an autoimmune reaction similar to Heparin-induced Thrombocytopenia (HIT), following administration of the AstraZeneca (AZ) and Johnson & Johnson/Janssen (JJ) vaccine. - Estimated rates are evolving. The reported incidence of 1/50000-1/100000 continues to evolve as milder cases are being increasingly recognized. The reaction has almost entirely occurred following the first administration of the vaccine, however, as many have not yet received a second AZ dose, the information continues to evolve. - Because a large proportion of individuals with VITT are presenting with Cerebral Venous Thrombosis (CVT), it is important that neurologists are aware of the condition and suggested treatment pathways. - Importantly, neurologists should:
- Clinical decision tool and current management recommendations from Thrombosis Canada (update 26 Apr) and the Ontario Science Table (update May 7) can be found here: |
What is the issue?
Since mid-March, hundreds of cases of atypical thromboses with thrombocytopenia have been reported in individuals who had received an adenovirus vector COVID vaccination (AstraZeneca [AZ] or Johnson&Johnson/Janssen [JJ]) within the past month. While cerebral venous thrombosis with or without involvement of additional sites has been the most commonly reported presentation, other cases have included splanchnic vein thrombosis, pulmonary embolism/DVT as well as arterial thromboembolic events.
The proposed mechanism, appears to be a reaction similar to heparin-induced thrombocytopenia (HIT) wherein antibodies directed against platelet factor 4 appear to be driving a response that leads to thrombocytopenia and, in most cases, thromboembolic events. Antibodies against platelet factor 4 have been reported across multiple case series of VITT, and from multiple labs. Anionic molecules besides heparin can bind to platelet factor 4, exposing an epitope against which antibodies are directed and triggering a reaction similar to HIT. The reaction has been shown in-vitro to be inhibited by IVIg, which binds to the Fc-gamma receptor on platelet factor 4.
Who is affected?
While initial reports suggested that individuals under the age of 60 and women in particular were likelier to be affected, emerging data suggests that all ages are affected and the sex distribution is more equitable than per early reports, though younger individuals and females comprise the majority of cases. A summary of recent case series of VITT-related CVT is included here:
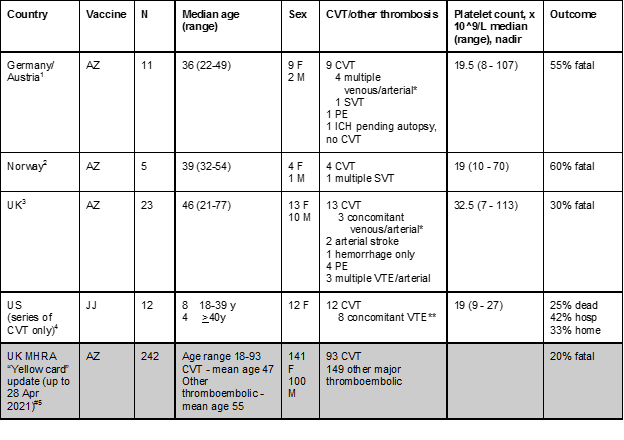
CVT - cerebral venous thrombosis; SVT - splanchnic vein thrombosis; PE - pulmonary embolism; VTE - venous thromboembolism; IJ - internal jugular; *Includes concomitant PE and/or SVT and/or arterial or diffuse microvascular involvement; **Includes IJ involvement, SVT, PE, DVT; # - “thrombosis and thrombocytopenia”; anti-PF4 results and platelet counts not included
What should we do as neurologists?
(0) Disseminate this information to your trainees, and neurology colleagues to spread awareness with respect to recognizing this entity and the distinct treatment strategy for this condition
(1) Recognize potential cases of vaccine-associated CVT before treatment is initiated;
- Obtain history related to recent COVID (or other) vaccination, including vaccine type, first or second dose, and where and when it was administered.
- Review the CBC to look for any presence of thrombocytopenia. Keep in mind that there are other potential causes (e.g. ITP, DIC, TTP/HUS, APLAS) that could potentially present with thrombocytopenia besides VITT.
- If giving phone/telestroke advice, ensure that the referring physician is aware of the importance of conveying complete and accurate information related to the above.
(2) Recognize the need for distinct management of VITT-associated CVT in the context of thrombocytopenia and engage hematology urgently.
Suspect VITT-ASSOCIATED CVT when:
- Confirmed diagnosis of CVT
- History of AZ/JJ COVID vaccination 4-28 days ago;
- Platelets < 150 x 10^9/L and elevated d-dimer
Suggested management considerations:
- Engage hematology/thrombosis service early for discussion of management of thrombocytopenia and anticoagulation
- For anticoagulation, a collaborative approach with hematology is appropriate, and risks and benefits of anticoagulation will need to be weighed carefully in the case of severe thrombocytopenia. Oral factor Xa inhibitors (apixaban, rivaroxaban or edoxaban) are the treatment of choice. IV argatroban may be a potential consideration when oral anticoagulation is not appropriate or not possible.
- Do not give platelet transfusions
- In collaboration with hematology, give IVIg 1 g/kg/d x 2 days.
- Prior to initiating IVIg, draw blood samples for HIT ELISA testing and special platelet immunology testing at the McMaster Platelet Immunology Lab (freeze a sample of serum and a sample of plasma). The hematology services are coordinating provincial pathways to forward samples to McMaster.
- Do not administer heparin, low-molecular weight heparin or other heparinoids, which may worsen the HIT-like response.
- Monitor for signs and symptoms for other venous thrombotic events, including splanchnic vein thrombosis and DVT/PE.
(3) Proactively report potential cases to local public health agencies as well as Health Canada
- Potential cases should be reported as soon as possible to your public health unit and to Health Canada. The Health Canada reporting form for vaccine-associated complications can be found here:
(4) Engage in standardized data collection to collate relevant information alongside our international colleagues to best identify any groups at particularly high risk of developing this condition.
- If you have a case of suspected VITT-CVT or other non-VITT CVT following any type of COVID vaccination, please contact our national VITT-CVT monitoring group (contact Vanessa Dizonno, vanessa.dizonno@ubc.ca). In collaboration with the International Cerebral Venous Thrombosis Research Consortium, our group is collecting clinical information about post-vaccination cases of CVT to better understand the risk factors, pathophysiology and clinical course of VITT- and non-VITT-associated CVT. Through the treating clinician, we are getting consent from patients/families for collection of routine clinical information.
Additional resources:
Thrombosis Canada guidance: https://thrombosiscanada.ca/covid-19/
AHA/ASA Guidance https://www.ahajournals.org/doi/pdf/10.1161/STROKEAHA.121.035564
References:
- Greinacher A, Thiele T, Warkentin TE, Weisser K, Kyrle PA, Eichinger S. Thrombotic Thrombocytopenia after ChAdOx1 nCov-19 Vaccination. N. Engl. J. Med. [Internet]. 2021;Available from: http://dx.doi.org/10.1056/NEJMoa2104840
- Schultz NH, Sørvoll IH, Michelsen AE, Munthe LA, Lund-Johansen F, Ahlen MT, Wiedmann M, Aamodt A-H, Skattør TH, Tjønnfjord GE, et al. Thrombosis and Thrombocytopenia after ChAdOx1 nCoV-19 Vaccination. N. Engl. J. Med. [Internet]. 2021;Available from: http://dx.doi.org/10.1056/NEJMoa2104882
- Scully M, Singh D, Lown R, Poles A, Solomon T, Levi M, Goldblatt D, Kotoucek P, Thomas W, Lester W. Pathologic Antibodies to Platelet Factor 4 after ChAdOx1 nCoV-19 Vaccination. N. Engl. J. Med. [Internet]. 2021;Available from: http://dx.doi.org/10.1056/NEJMoa2105385
- See I, Su JR, Lale A, Woo EJ, Guh AY, Shimabukuro TT, Streiff MB, Rao AK, Wheeler AP, Beavers SF, et al. US Case Reports of Cerebral Venous Sinus Thrombosis With Thrombocytopenia After Ad26.COV2.S Vaccination, March 2 to April 21, 2021 [Internet]. JAMA. 2021;Available from: http://dx.doi.org/10.1001/jama.2021.7517
- Coronavirus vaccine - weekly summary of Yellow Card reporting [Internet]. [cited 2021 May 8];Available from: https://www.gov.uk/government/publications/coronavirus-covid-19-vaccine-adverse-reactions/coronavirus-vaccine-summary-of-yellow-card-reporting


